|
3/31/2021 0 Comments Lewis Structure Generator
Send feedback Visit WolframAlpha SHARE Email Twitter Facebook Share via Facebook More.
Lewis Structure Code Below IntoShare This Page Digg StumbleUpon Delicious Reddit Blogger Google Buzz Wordpress Live TypePad Tumblr MySpace LinkedIn URL EMBED Make your selections below, then copy and paste the code below into your HTML source.

Lewis Structure Install The WolframAlphaTo embed a widget in your blogs sidebar, install the WolframAlpha Widget Sidebar Plugin, and copy and paste the Widget ID below into the id field. To add a widget to a MediaWiki site, the wiki must have the Widgets Extension installed, as well as the code for the WolframAlpha widget. To include the widget in a wiki page, paste the code below into the page source. This may take up to a minute, depending on your location and the connection. Lewis Structure Free Molecule EditorBienfait and P. Ertl, JSME: a free molecule editor in JavaScript, Journal of Cheminformatics 5:24 (2013) article. Search for: Organic Chemistry 1 and 2 Summary Sheets Ace your Exam Chemistry Steps LLC Organic Chemistry Study Materials, Practice Problems, Summary Sheet Guides, Multiple-Choice Quizzes. This is why it is essential to able to convert Lewis and condensed structures to bond-line representation. Lets first quickly summarize what Bond-Line (Zig-zag) structures are: To interpret a bond-line structure, assume that: 1) There is a carbon at each junction (corner) and periphery. Each carbon has enough hydrogens to have 4 bonds (unless there is a formal charge) So, essentially in bond-line structures, we dont show the hydrogens and carbons. All the heteroatoms (any atoms except Carbon) are shown and the hydrogens on these heteroatoms are shown as well. Converting a Condensed Structure to a Bond-line Now, lets say you need to convert the following condensed structure into a bond-line structure: There is no formula or best approach for doing this, but we will follow the following strategy that will eliminate any possibility of making a mistake when converting to a bond-line structure. Note: Numbering atoms will help you immensely for doing any transformation in Organic Chemistry. It does not have to be in a particular order or following the IUPAC rules. Simply connect all the atoms from left to right. Follow the table for standard valencies on the bottom-left to identify the wrong connections: We can identify the following sections with incorrect valency of elements: 3) Rearrange the atoms such that they have the correct number of bonds: (It will be very beneficial to learn the functional groups at this point) These are the only ways to have the correct number of bonds and lone pairs for the atoms in red boxes. Knowing the functional groups will make it much easier to put the atoms in the right places. Draw the carbon chain in a zig-zag form. Putting the first atom up or down doesnt matter as long as you keep the correct connectivity of atoms. Erase the carbon atoms together with the hydrogens on them. Keep all the heteroatoms (any atom except carbon) together with the hydrogens on them. The table below shows some of the functional groups that often lead to a wrong connectivity pattern as it is not obvious to put the atoms correctly right from beginning: Once again, knowing the functional groups at this point will help you tremendously when converting Lewis or condensed structures to bond-line structures. 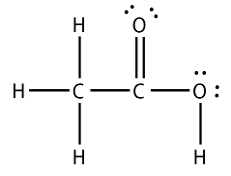
Username Password Remember Me Forgot Password Solution This content is for registered users only. Convert the following condensed structure into Bond-line structure: (CH 3 ) 2 CHCH 2 OCH 2 CHNC(CH 3 ) 3 Check your answers This content is for registered users only. Convert the following condensed structure into Bond-line structure: CH 3 (CH 2 ) 2 CONHCH 2 CH(CH 3 )OCH 3 Check your answers This content is for registered users only. Convert the following condensed structure into Bond-line structure: CH 3 COOCH 2 (CH) 2 CCCH 3 Check your answers This content is for registered users only. Convert the following condensed structure into Bond-line structure: CH 2 CHCH 2 NHCH 2 COOCH 3 Check your answers This content is for registered users only. Take a Quiz Now Check Also: Bond-Line or Skeletal Structures Functional Groups in Organic Chemistry Curved Arrows Resonance Structures in Organic Chemistry Assessing the Relative Importance of Resonance Structures Drawing Complex Patterns in Resonance Structures Localized and Delocalized Lone Pairs Leave a Comment Cancel reply Comment Name Email Website Notify me of followup comments via e-mail.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
